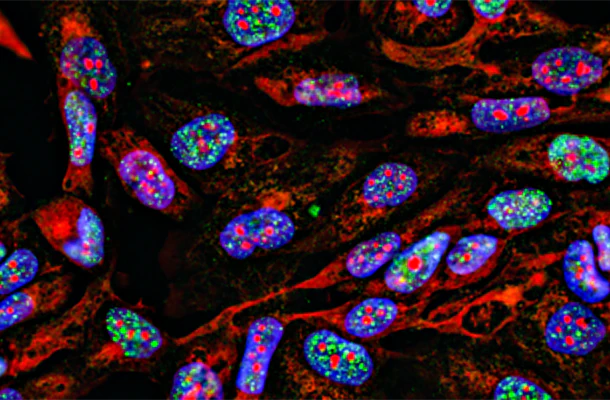
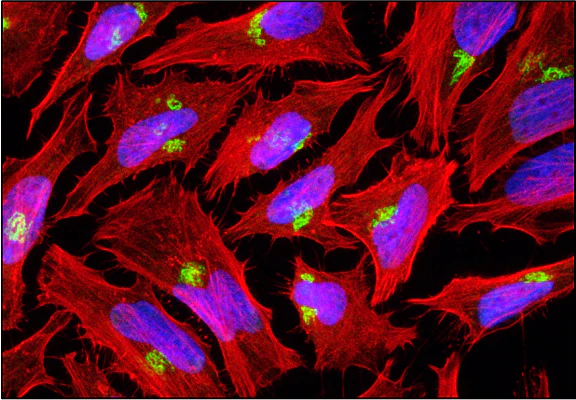
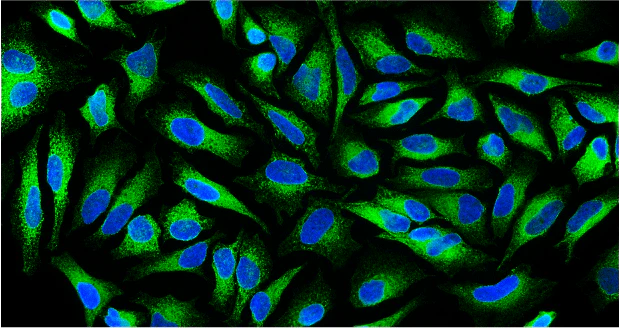
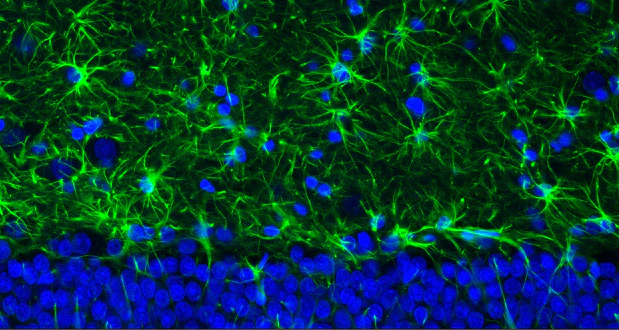
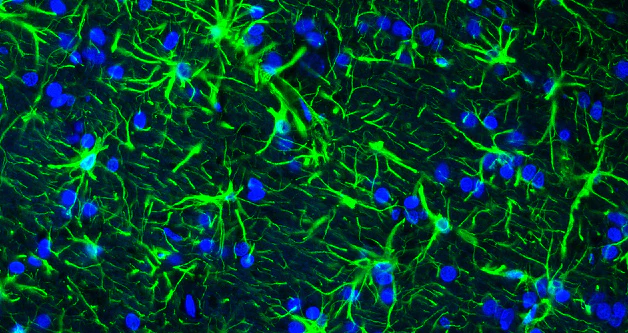
UEA-I-Biotin, Conjugated
The anti-H (O) hemagglutinating activity of Ulex europaeus has been used widely to confirm blood group O activity. UEA I binds to many glycoproteins and glycolipids containing α –linked fucose residue. The purified lectin appears to be a dimer of two distinct polypeptide chains associated by noncovalent forces. This lectin does not react with Lea active blood group substance. The native protein has pI=6.0-6.1 and exhibits a Molecular weight 60,000-68,000 during gel filtration on Sephadex column.
Product Information
Product Information
Shipping & Returns
Shipping & Returns
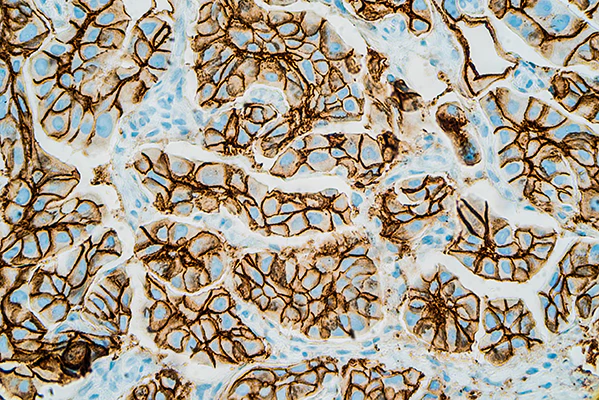
UEA-I-Biotin, Conjugated
UEA-I-Biotin, Conjugated
The anti-H (O) hemagglutinating activity of Ulex europaeus has been used widely to confirm blood group O activity. UEA I binds to many glycoproteins and glycolipids containing α –linked fucose residue. The purified lectin appears to be a dimer of two distinct polypeptide chains associated by noncovalent forces. This lectin does not react with Lea active blood group substance. The native protein has pI=6.0-6.1 and exhibits a Molecular weight 60,000-68,000 during gel filtration on Sephadex column.
Product Information
Product Information
Shipping & Returns
Shipping & Returns
Description
The anti-H (O) hemagglutinating activity of Ulex europaeus has been used widely to confirm blood group O activity. UEA I binds to many glycoproteins and glycolipids containing α –linked fucose residue. The purified lectin appears to be a dimer of two distinct polypeptide chains associated by noncovalent forces. This lectin does not react with Lea active blood group substance. The native protein has pI=6.0-6.1 and exhibits a Molecular weight 60,000-68,000 during gel filtration on Sephadex column.