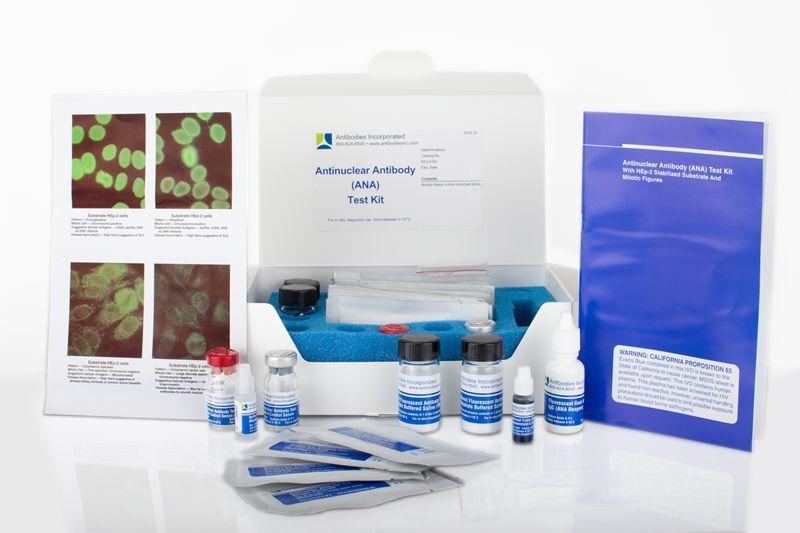
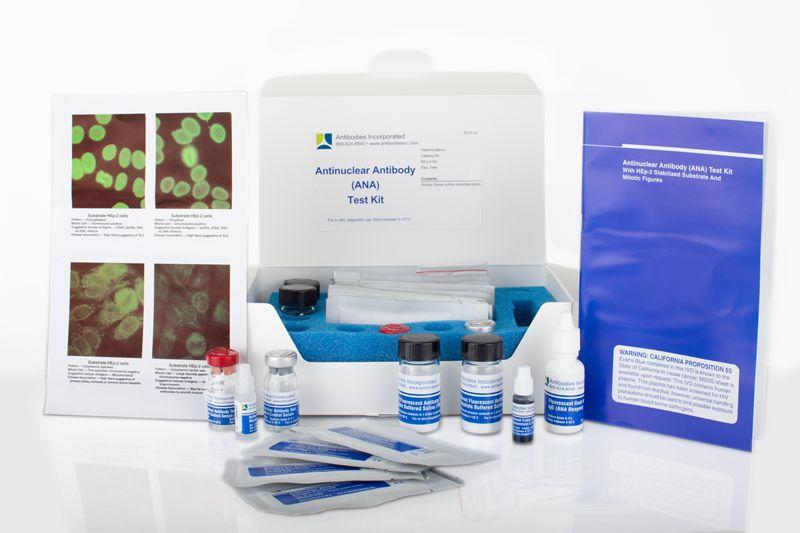
ANA Positive Control (Homogeneous) In-Vitro Diagnostic Kit
Our ANA Positive Control (Homogeneous) in-vitro diagnostic kit from Antibodies Incorporated is an FDA-approved IVD kit used to aid in the diagnosis of systemic rheumatic diseases. This Anti-Nuclear Antibody IVD kit and all components are produced in-house.
Product Information
Product Information
Shipping & Returns
Shipping & Returns

ANA Positive Control (Homogeneous) In-Vitro Diagnostic Kit
ANA Positive Control (Homogeneous) In-Vitro Diagnostic Kit
Our ANA Positive Control (Homogeneous) in-vitro diagnostic kit from Antibodies Incorporated is an FDA-approved IVD kit used to aid in the diagnosis of systemic rheumatic diseases. This Anti-Nuclear Antibody IVD kit and all components are produced in-house.
Product Information
Product Information
Shipping & Returns
Shipping & Returns
Description
Our ANA Positive Control (Homogeneous) in-vitro diagnostic kit from Antibodies Incorporated is an FDA-approved IVD kit used to aid in the diagnosis of systemic rheumatic diseases. This Anti-Nuclear Antibody IVD kit and all components are produced in-house.